Factor V Leiden Test:
Why Factor - V (Leiden) Test ?
CLINICAL INFORMATION
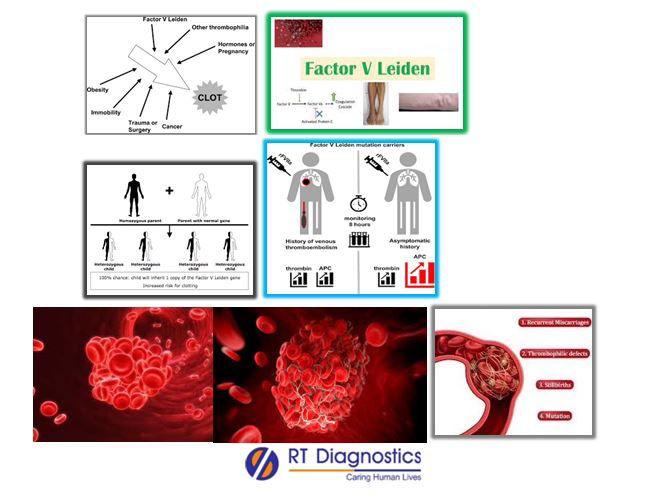
Haemophilia is a disease where blood does not clot properly. Each time a person bleeds, it triggers a series of reactions known as the coagulation cascade to form clots to stop bleeding. Clotting factor tests are also called ‘factor assays tests’ or “screening for factor assays” are required to diagnose bleeding disorders. Other screening tests for detecting clotting disorders include CBC (looks for any abnormalities in RBCs, WBCs and mainly the abnormalities seen platelets and in the platelet count), activated partial thromboplastin time (APTT): These tests look for how long it takes for blood to clot and hence if any of these clotting factors are too low then clotting will take longer time, Prothrombin time (PT): It measures the time taken to form a clot as it measures the clotting ability of factors I, II, V, VII and X and thus if any of these clotting factors are too low then clotting will take longer time, Fibrinogen test is also known as Factor 1 Test: This test is performed when the patient has abnormal PT or APTT test results, clotting factor tests also known as Factor Assays: This test assay helps to screen for diagnosis of bleeding disorder as in case of haemophilia and also to assess its severity (i.e this test helps inaccurate classification of haemophilia) etc. Patients before this test must not have received medications such as Coumadin, warfarin, blood thinners, heparin therapy like LMWH etc, since it will interfere with the true test results. Other factors which may influence the test results are vitamin K deficiency and conditions like liver diseases, lupus anticoagulants etc. Factor V is also called Leiden factor because it was originally discovered at the University of Leiden (Netherlands), hence known also as“Factor V Leiden - FVL”, ‘Leiden Clotting Factor’ etc. Factor V Leiden thrombophilia is an inherited disorder of blood clotting. It is associated with an increased risk of complications related to pregnancies such as miscarriages, preeclampsia, placental abruption, slow fetal growth etc. This defect can arise due to qualitative defect or quantitative defect of this clotting factor V leading to hyper-coagulable disorder. Usually, factor V is inactivated by activated protein C – APC (thus also called APC resistance or Activated Protein C - Resistance), but when factor V Leiden genetic mutation occurs the altered factor V protein resists inactivation by APC, therefore resulting in abnormal clots. It is associated with a mutation of one of the clotting factors in the blood, and hence this mutation increases the chance of developing abnormal blood clots in the lungs (pulmonary embolism), legs (DVT) and/or infarcts due to embolism can occur in the blood supply (venous thromboembolism - VTE) to the liver (hepatic vein), kidneys (renal vein), intestine (mesenteric supply), brain (cerebral vein), eyes etc. Therefore these patients are associated with long term health problems or their complications can be life-threatening. Risk factors include – smoking, immobility, OCPs with estrogen, patients with inborn errors of metabolism with elevated levels of homocysteine, surgeries or injuries etc. Clinical manifestations of this disorder present with signs and symptoms of DVT, sudden shortness of breath, chest pain while breathing, rapid heartbeat, bloody sputum etc.
Factor IX activity test: Factor Vis Leiden test is performed in suspected patients with abnormal clots. This test is indicated by a physician when the patient had episodes of abnormal clotting history in the past following injuries and/or if the patient has a strong family genetic history of abnormal blood clots (thrombotic episodes).
Additional tests include lupus anticoagulant, protein C, Protein S, Antithrombin, Homocysteine, Factor V gene R2 mutation, Methylenetetrahydrofolate reductase (MTHFR) gene mutation etc. Other tests include stool tests and urinalysis, genetic testing etc.
General Instructions:
Sample Requirement: Specimen - Blood sample collected from the vein. Test Preparation: None.
NOTE - Sample for specimen collections may vary based on the patient’s condition/cases according to the patient’s presenting complaints/signs or symptoms:
SPECIMEN REQUIREMENT (Special or Rare Cases) - As instructed and guided by Physician / Clinician / Pathologist / as per Laboratory’s requirements, according to procedures and protocols.
This Multi-Specialty Clinical Referral Laboratory RT DIAGNOSTICS provides precise and accurate tests with an extensive range of testing services to the medical centres to help in the diagnosis and identification of pathology in the test specimens for infectious diseases and also to evaluate the function of organ systems of the patient. It prevents further complications and helps to stabilize and restore health to near normalcy at the earliest without delay.



